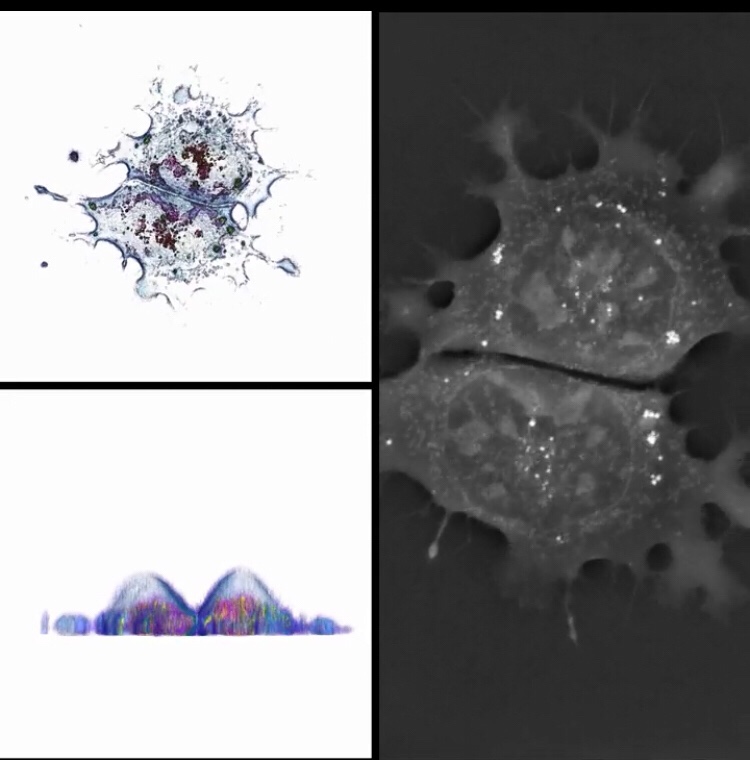
Artificial Intelligence with Deep Learning for Medical Records and Images
Brigham and Women's to apply deep learning to medical records and images Hospitals say the AI method is in use today automating things humans do well but don't have the time for – and there's much more to come. Read on: www.healthcareitnews.com/news/mass-general-brigham-and-womens-apply-deep-learning-medical-records-and-images